A brief overview of aerosol fire extinguishing systems and fire extinguishing classes
The aerosol cools the environment after evacuation, so the room temperature decreases due to the physical effect of the aerosols.
In other words, the fire cannot explode while the aerosol gas is completely flowing into the area.
There is a new product that can provide a system for effective fire protection in the new generation of firefighters. With fire extinguishers, airborne particles with strong fire extinguishing function, completely environmentally friendly, cost-effective, which includes the solid form of potassium salts, is completely safe in terms of health and environment. It also does not require pressurized tanks or pressure vessels and also does not require 100% non-leakage conditions in the environment.
New, clean and ecological
AEROSOL fire extinguishing technology
There is a new product that can provide a system for effective fire protection in the new generation of firefighters. Air-Particle Aerosol Fire Extinguishing System with fire-extinguishing function, fully environmentally friendly, cost-effective, containing solid form of potassium salts, completely harmless to health and the environment. It also does not require pressurized tanks or pressure vessels and also does not require 100% non-leakage conditions in the environment.
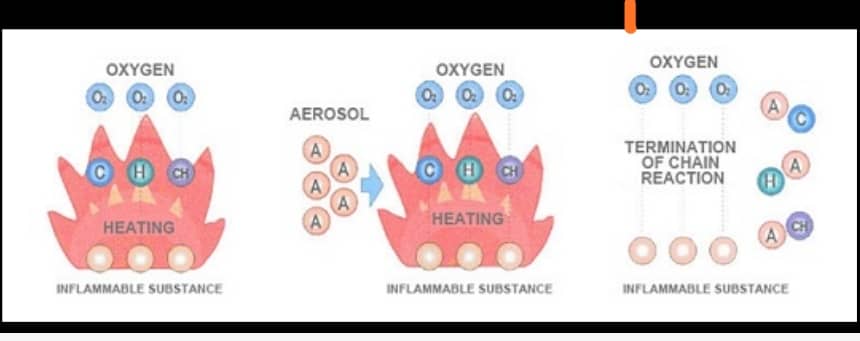
How does an aerosol particle extinguisher work?
If we want to define the word “airborne particles”: in short, it is a multi-phase system, which is formed by the scattering of very small solid or liquid particles and its size is less than 10 microns in a medium consisting of Is gas. This system is a multi-phase suspended volume in which it fills the volume for a long time without settling.
Basically, aerosol extinguishers are contained inside metal cans as a compressed block of solid compounds produced with alkali metal salts and other auxiliary chemicals. Fire detection systems create these solid chemical compound blocks in the event of a fire.
When these solid composite blocks are activated manually or automatically by the signal of the control panel of the fire extinguishing panel and the extinguisher to the excitation device, which is located in the metal housing of the airborne particle fire extinguishers. Therefore, this signal initiates a redox reaction (reduction of oxidation) inside the uncompressed chamber, causing an exciting fog and a very dense fire.
The fog contains potassium carbonate and bicarbonate with a micron size of 2 microns, water vapor, carbon dioxide and nitrogen gas.
It has physical properties (cooling) and chemical extinguisher in fire.
The large surface area and large amount of small solid aerosol particles cause physical extinction. It is also chemically formed by the use of stable unsuitable chemicals (free radicals) that are generated by the heat of reaction of fire and also feed on combustion, and these free radicals (hydrogen and hydroxyl compounds) as fuel in the fire and After evacuating the aerosol, the gas of these free radicals reacts with the gas components of airborne particles, and these gas components of airborne particles absorb these fire fuels and prevent free radicals from being replaced by a fire reaction. Do not let the fire extinguish the aerosol with a chemical effect.
Compared to the 1230 and FM200 tip gases, which replace the halon gases, the aerosol fire extinguisher is used as the most efficient and cost-effective.


